Kesum
|
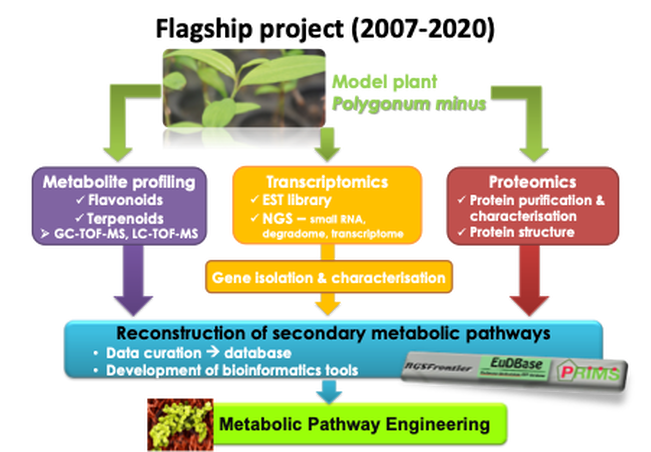
Kesum (Polygonum minus syn. Persicaria minor) is an herbal plant in the Polygonaceae family which is rich in ethnomedicinal plants. It grows in Southeast Asian countries and traditionally used as medicine. This plant produces diverse secondary metabolites such as phenolic compounds and their derivatives, which are known to have roles in plant abiotic and biotic stress responses. Its sesquiterpenes contribute to its pungent flavour and fragrance in local cooking, known as laksa.
|
Studies
Transcriptomics
|
The chemical composition and characteristic pungent fragrance of Polygonum minus have been extensively studied due to its culinary and medicinal properties. There are only a few transcriptome sequences available for species from this important family of medicinal plants. The limited genetic information from the public expressed sequences tag (EST) library hinders further study on molecular mechanisms underlying secondary metabolite production.
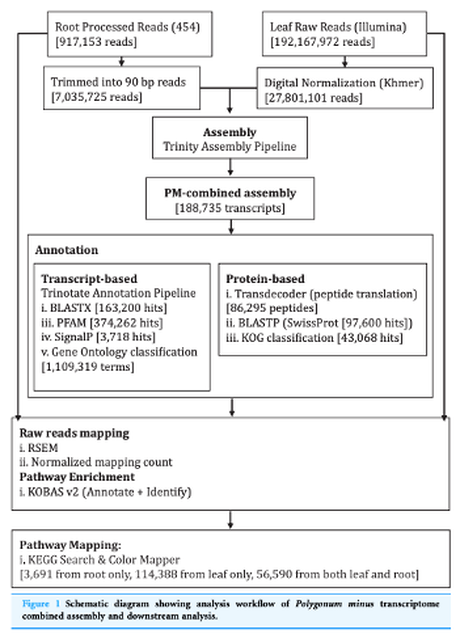
We performed a hybrid assembly of 454 and Illumina sequencing reads from Polygonum minus root and leaf tissues, respectively, to generate a combined transcriptome library as a reference. A total of 34.37 million filtered and normalized reads were assembled into 188,735 transcripts with a total length of 136.67 Mbp. We performed a similarity search against all the publicly available genome sequences and found similarity matches for 163,200 (86.5%) of Polygonum minus transcripts, largely from Arabidopsis thaliana (58.9%). Transcript abundance in the leaf and root tissues were estimated and validated through RT-qPCR of seven selected transcripts involved in the biosynthesis of phenylpropanoids and flavonoids. All the transcripts were annotated against KEGG pathways to profile transcripts related to the biosynthesis of secondary metabolites. |
Significance
We described the first comprehensive transcriptome profile of Polygonum minus leaf and root tissues for the curation of secondary metabolite-related transcripts. We compared the annotation results of three Polygonum minus assemblies (leaf, root, and combined) with that of previous EST library. This comparison provides a useful resource for gene discovery using Polygonum minus combined assembly. We have established a reference transcriptome profile of Polygonum minus with annotations of transcript descriptions for future investigation on specific processes or pathways, especially on the effect of environmental stresses and the biosynthesis of secondary metabolites. We also identified some of the enriched metabolite pathways in the root and leaf tissues. The identification of transcripts related to secondary metabolite biosynthesis will aid in further exploitation of the genetic resource from this herbal plant for future biotechnological development. This comprehensive transcriptome profile will serve as a useful sequence resource for molecular genetics and evolutionary research on secondary metabolite biosynthesis in Polygonaceae family.
|
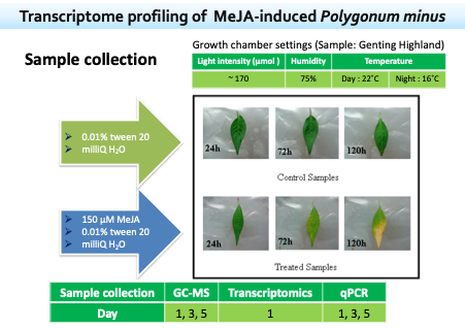
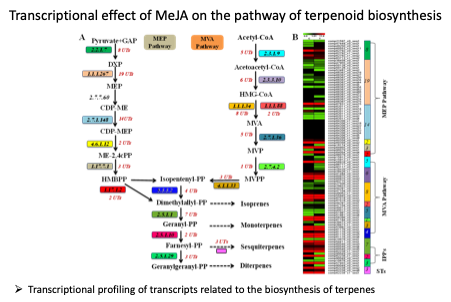
Methyl jasmonate (MeJA) is a plant signaling molecule that triggers transcriptional reprogramming in secondary metabolism and activation of defense responses against many biotic and abiotic stresses. However, the effect of MeJA elicitation on the genome-wide expression profile in the leaf tissue of P. minus has not been well-studied due to the limited genetic information.
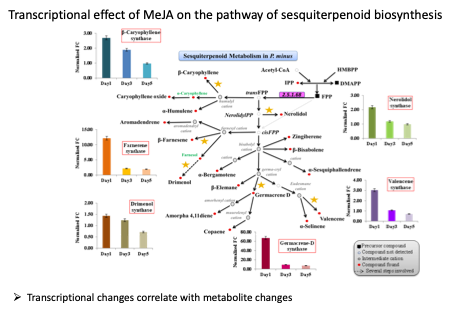
Hence, we performed Illumina paired-end RNA-seq for de novo reconstruction of P. minus leaf transcriptome to identify differentially expressed genes (DEGs) in response to MeJA elicitation. A total of 182,111 unique transcripts (UTs) were obtained by de novo assembly of 191.57 million paired-end clean reads using Trinity analysis pipeline. A total of 2,374 UTs were identified to be significantly up-/down-regulated 24 h after MeJA treatment. |
These UTs comprising many genes related to plant secondary metabolite biosynthesis, defense and stress responses. To validate our sequencing results, we analyzed the expression of 21 selected DEGs by quantitative real-time PCR and found a good correlation between the two analyses.
The single time-point analysis in this work not only provides a useful genomic resource for P. minus but also gives insights on molecular mechanisms of stress responses in P. minus.
The single time-point analysis in this work not only provides a useful genomic resource for P. minus but also gives insights on molecular mechanisms of stress responses in P. minus.
Significance
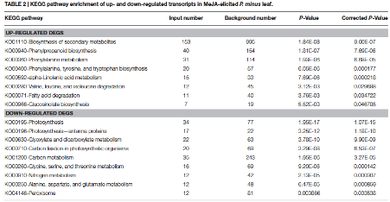
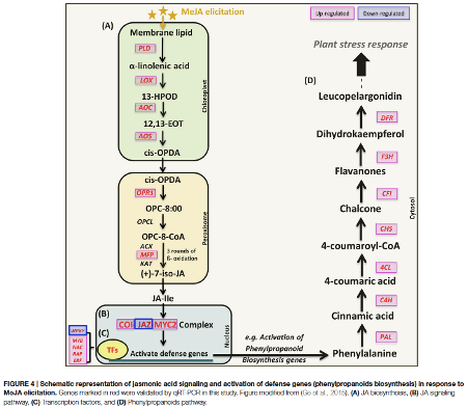
In this study of MeJA-elicited P. minus leaf transcriptome, 182,111 UTs were assembled with 2,374 DEGs identified. The is the first RNA-seq study on the response to MeJA of a non-model plant from Polygonaceae family, which comprises many useful medicinal plants. Functional annotation of DEGs based on COG, GO and KEGG showed the upregulation of UTs involve in plant defense and protective functions, as well as secondary metabolic processes. Conversely, UTs related to photosynthesis were down-regulated. This is in accordance to the TF families found to be differentially expressed. MeJA elicitation is found to upregulate endogenous JA biosynthesis. The molecular mechanism on how MeJA triggers the upregulation of phenylpropanoid pathway is proposed and validated by RT-qPCR analysis. These results support the physiological trade-offs between stress response and growth despite that the underlying mechanism remains unclear. Furthermore, current P. minus transcriptome provides an extensive sequence resource for gene discovery and functional studies in related species.
In this study of MeJA-elicited P. minus leaf transcriptome, 182,111 UTs were assembled with 2,374 DEGs identified. The is the first RNA-seq study on the response to MeJA of a non-model plant from Polygonaceae family, which comprises many useful medicinal plants. Functional annotation of DEGs based on COG, GO and KEGG showed the upregulation of UTs involve in plant defense and protective functions, as well as secondary metabolic processes. Conversely, UTs related to photosynthesis were down-regulated. This is in accordance to the TF families found to be differentially expressed. MeJA elicitation is found to upregulate endogenous JA biosynthesis. The molecular mechanism on how MeJA triggers the upregulation of phenylpropanoid pathway is proposed and validated by RT-qPCR analysis. These results support the physiological trade-offs between stress response and growth despite that the underlying mechanism remains unclear. Furthermore, current P. minus transcriptome provides an extensive sequence resource for gene discovery and functional studies in related species.
Proteomics
|
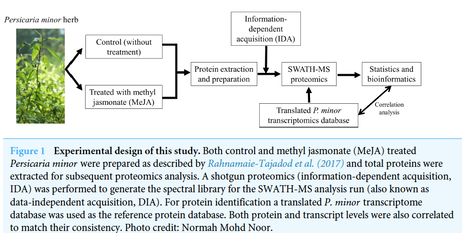
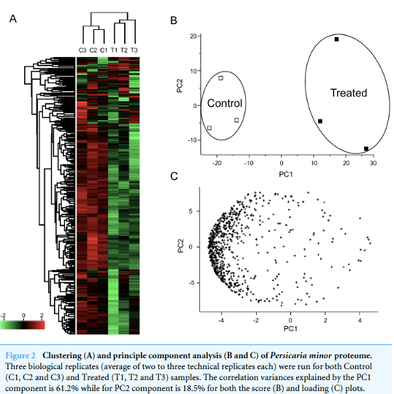
Jasmonic acid (JA) and its derivative, methyl JA (MeJA) are hormonal cues released by plants that signal defense response to curb damages from biotic and abiotic stresses. To study such response, a tropical herbal plant, Persicaria minor, which possesses pungent smell and various bioactivities including antimicrobial and anticancer, was treated with MeJA. Such elicitation has been performed in hairy root cultures and plants such as Arabidopsis and rice, yet how MeJA influenced the proteome of an herbal species like P. minor is unknown.
P. minor plants were exogenously elicited with MeJA and leaf samples were subjected to SWATH-MS proteomics analysis. A previously published translated transcriptome database was used as a reference proteome database for a comprehensive protein sequence catalogue and to compare their differential expression. |
|
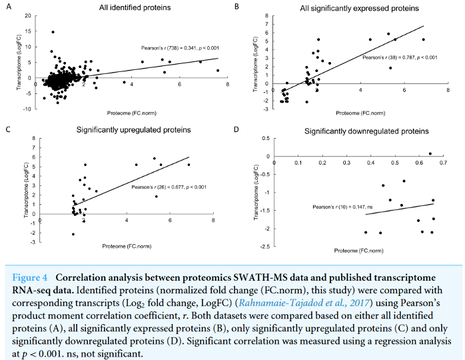
From this proteomics informed by transcriptomics approach, 751 proteins were successfully profiled of which 40 proteins were significantly different between control and MeJA-treated samples. Furthermore, a correlation analysis between both proteome and the transcriptome data sets suggests that significantly upregulated proteins were positively correlated with their cognate transcripts (Pearson’s r = 0.677) while a weak correlation was observed for downregulated proteins (r = 0.147).
|
MeJA treatment induced the upregulation of proteins involved in various biochemical pathways including stress response mechanism, lipid metabolism, secondary metabolite production, DNA degradation and cell wall degradation. Conversely, proteins involved in energy expensive reactions such as photosynthesis, protein synthesis and structure were significantly downregulated upon MeJA elicitation. Overall protein-transcript correlation was also weak (r = 0.341) suggesting the existence of post-transcriptional regulation during such stress. In conclusion, proteomics analysis using SWATH-MS analysis supplemented by the transcriptome database allows comprehensive protein profiling of this non-model herbal species upon MeJA treatment.
Significance
This is the first proteomics study on this non-model herbal species, P. minor particularly using an advanced proteomics platform called SWATH-MS, enriched with a previously reported transcriptome database for protein identification. This has enabled us to profile a comprehensive proteome coverage of 751 proteins, and forty were found differentially expressed. The modulated levels of these proteins suggest that the hormone invoked defense and recovery response but suppressed proteins involved in growth and development. Correlation analysis between our proteome study and previously reported transcriptome analysis suggests that post-transcriptional and post-translational regulation may have existed to regulate certain groups of proteins.
This is the first proteomics study on this non-model herbal species, P. minor particularly using an advanced proteomics platform called SWATH-MS, enriched with a previously reported transcriptome database for protein identification. This has enabled us to profile a comprehensive proteome coverage of 751 proteins, and forty were found differentially expressed. The modulated levels of these proteins suggest that the hormone invoked defense and recovery response but suppressed proteins involved in growth and development. Correlation analysis between our proteome study and previously reported transcriptome analysis suggests that post-transcriptional and post-translational regulation may have existed to regulate certain groups of proteins.
Metabolomics
|
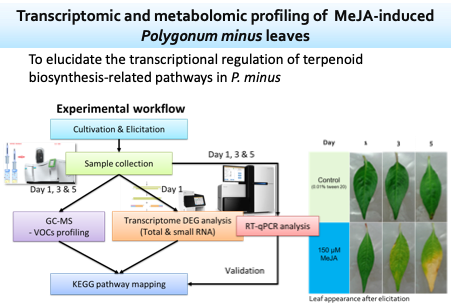
Polygonum minus Huds. is a medicinal aromatic plant rich in terpenes, aldehydes, and phenolic compounds. Methyl jasmonate (MeJA) is a plant signaling molecule commonly applied to elicit stress responses to produce plant secondary metabolites. In this study, the effects of exogenous MeJA treatment on the composition of volatile organic compounds (VOCs) in P. minus leaves were investigated by using a metabolomic approach. Time-course changes in the leaf composition of VOCs on days 1, 3, and 5 after MeJA treatment were analyzed through solid-phase microextraction (SPME) and gas chromatography-mass spectrometry (GC-MS).
|
Our results suggest that MeJA enhances the activity of biosynthetic pathways for aldehydes and terpenes in P. minus. Hence, the production of aromatic compounds in this medicinal herb can be increased by MeJA elicitation. Furthermore, the relationship between MeJA elicitation and terpene biosynthesis in P. minus was shown through SPME-GC-MS analysis of VOCs combined with transcriptomic analysis of MeJA-elicited P. minus leaves from our previous study.
Significance
This is the first study of MeJA-induced volatiles in P. minus leaves using an integrated metabolomic and transcriptomic approach, which leads us to a better understanding of exogenous MeJA elicitation on the production of P. minus leaf volatiles. The SPME-GC–MS study with multivariate statistical analysis illustrated time-course changes in VOCs that occurred after elicitation via data clustering. Our results indicate that MeJA elicitation appears to mainly affect the synthesis of aldehydes and sesquiterpenes compared to other classes of volatiles.
Therefore, the MeJA induction of volatiles in P. minus provides an opportunity for the discovery of genes involved in the accumulation of induced terpenes and GLVs, which are important for the characteristic pungent smell of P. minus. The present study also opens an avenue to conduct further investigations into gene-metabolite networks, targeted enzyme analysis, and biotechnological applications.
This is the first study of MeJA-induced volatiles in P. minus leaves using an integrated metabolomic and transcriptomic approach, which leads us to a better understanding of exogenous MeJA elicitation on the production of P. minus leaf volatiles. The SPME-GC–MS study with multivariate statistical analysis illustrated time-course changes in VOCs that occurred after elicitation via data clustering. Our results indicate that MeJA elicitation appears to mainly affect the synthesis of aldehydes and sesquiterpenes compared to other classes of volatiles.
Therefore, the MeJA induction of volatiles in P. minus provides an opportunity for the discovery of genes involved in the accumulation of induced terpenes and GLVs, which are important for the characteristic pungent smell of P. minus. The present study also opens an avenue to conduct further investigations into gene-metabolite networks, targeted enzyme analysis, and biotechnological applications.
GC-MS/Olfactometric characterisation and aroma extraction dilution analysis of aroma active compounds in Polygonum minus essential oil
Polygonum minus Huds. is an aromatic plant with potential anti-inflammatory, anti-ulcer and diuretic properties. Although the volatile compounds from the essential oil of P. minus have been extensively studied, knowledge of the aroma-active compounds is still incomplete.
Polygonum minus Huds. is an aromatic plant with potential anti-inflammatory, anti-ulcer and diuretic properties. Although the volatile compounds from the essential oil of P. minus have been extensively studied, knowledge of the aroma-active compounds is still incomplete.
|
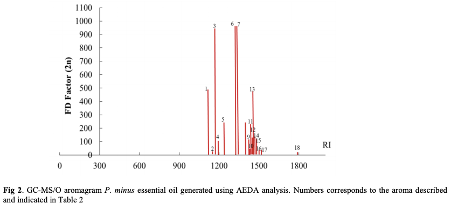
This study is aimed to investigate and to characterise the aroma-active compounds that contribute to the potent odour of P. minus. Essential oil was extracted from the leaves of kesum using hydrodistillation and was analysed by gas chromatography-mass spectrometry (GC-MS) to identify the volatile constituents. The important aroma-active compounds were further analysed by GC-MS/Olfactometry (GC-MS/O) and aroma extraction dilution analysis (AEDA).
|
|
The GC-MS analysis using Kováts retention indices detected 39 volatile compounds in the essential oil. Aldehydes represented the major class of compounds, encompassing 75.43% of the total peak area, followed by sesquiterpenes (13.19%), alcohols (8.07%) and organic acids (0.83%). The dominant components of the kesum essential oil were dodecanal (54.25%) and decanal (18.27%) in the aldehyde group. The sesquiterpenes that were detected include β-farnesene (1.74%), α-caryophyllene (1.72%), β-caryophyllene oxide (1.51%) and βselinene (1.36%). In this study, GC-MS/O and AEDA analysis identified dodecanal, decanal, farnesol, 1-nonanal and α-bergamotene as the main contributors to the characteristic fragrance of this plant.
|
Significance
This is the first olfactometric study on the essential oil of P. minus, and it is important for our understanding of the biosynthesis of volatile compounds in this medicinal herb. Moreover, the characterisation of aromatic compounds that contributed to the potent odour of P. minus can be further applied in the flavour and fragrance industries.
This is the first olfactometric study on the essential oil of P. minus, and it is important for our understanding of the biosynthesis of volatile compounds in this medicinal herb. Moreover, the characterisation of aromatic compounds that contributed to the potent odour of P. minus can be further applied in the flavour and fragrance industries.
Metabolite profiling reveals temperature effects on the VOCs and flavonoids of different plant populations
Temperature is one of the key factors in limiting the distribution of plants and controlling major metabolic processes. A series of simulated reciprocal transplant experiments were performed to investigate the effect of temperature on plant chemical composition. Polygonum minus of different lowland and highland origin were grown under a controlled environment with different temperature regimes to study the effects on secondary metabolites. We applied gas chromatography–mass spectrometry and liquid chromatography time-of-flight mass spectrometry to identify the chemical compounds.
Temperature is one of the key factors in limiting the distribution of plants and controlling major metabolic processes. A series of simulated reciprocal transplant experiments were performed to investigate the effect of temperature on plant chemical composition. Polygonum minus of different lowland and highland origin were grown under a controlled environment with different temperature regimes to study the effects on secondary metabolites. We applied gas chromatography–mass spectrometry and liquid chromatography time-of-flight mass spectrometry to identify the chemical compounds.
|
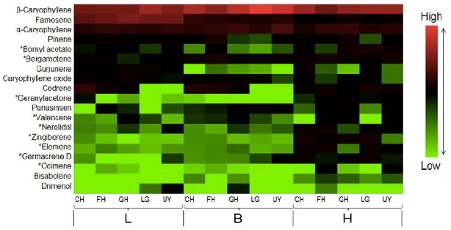
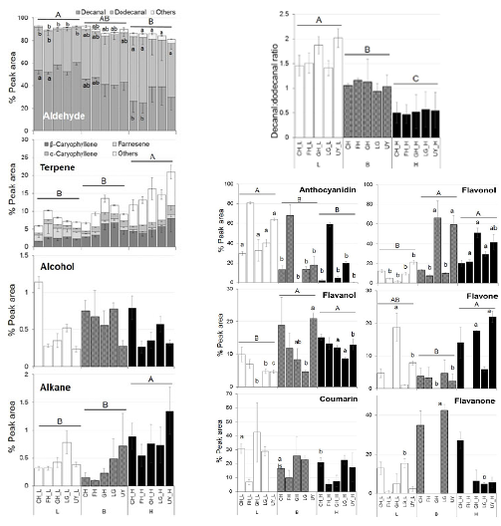
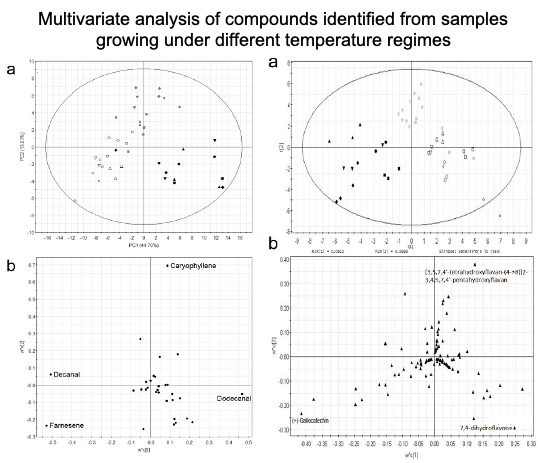
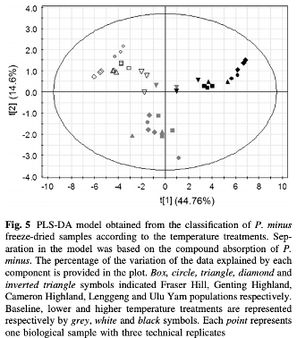
A total of 37 volatile organic compounds and 85 flavonoids were detected, with the largest response observed in the compositional changes of aldehydes and terpenes in highland plants under higher temperature treatment. Significantly less anthocyanidin compounds and larger amounts of flavonols were detected under higher temperature treatment. We also studied natural variation in the different plant populations growing under the same environment and identified compounds unique to each population through metabolite fingerprinting. This study shows that the origin of different plant populations influences the effects of temperature on chemical composition.
|
Significance
The results obtained from this study showed the variations in plant metabolism at different temperatures, with distinct accumulation of flavonoid and terpene compounds in response to different temperature treatments, which also depends on the origin of the different plant populations. Metabolites that were associated with higher temperature treatment were identified from PLS-DA loading plots, majority of which were assigned to aldehydes and terpenes. The metabolite fingerprinting approach detects shifts in the metabolome of P. minus in response to different temperature treatments. The content of flavonols and flavanols at higher temperature treatment were greater compared to lower temperature, perhaps to counteract a greater oxidative stress under suboptimal higher temperature. The difference observed when comparing the highland and lowland populations might reflect the different adaptive abilities of different populations to their natural growing temperatures. Unique compounds identified from each population can serve as metabolite fingerprints.
This study demonstrated that temperature had greater effects on the composition of VOCs than flavonoids, which was varied according to different populations. This knowledge is useful to inform cultivation conditions and choice of P. minus population to acquire plants producing desired flavonoid compounds. Furthermore, this study also implies that the effect of climate change on natural plant populations will be significantly varied and modelling of plant responses should include biogeographical distribution.
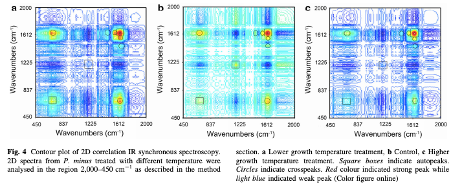
Direct discrimination of different plant populations and study on temperature effects by Fourier transform infrared spectroscopy
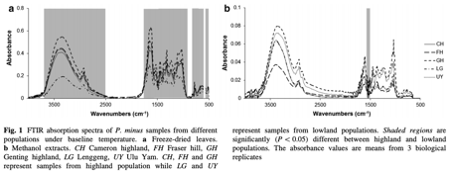
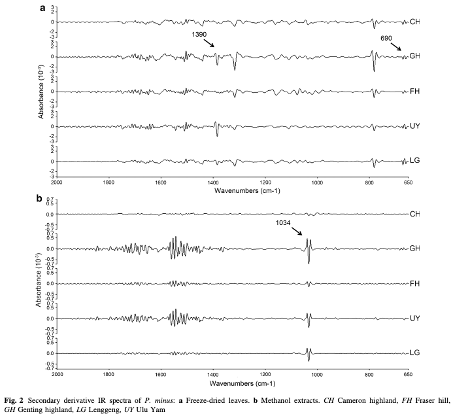
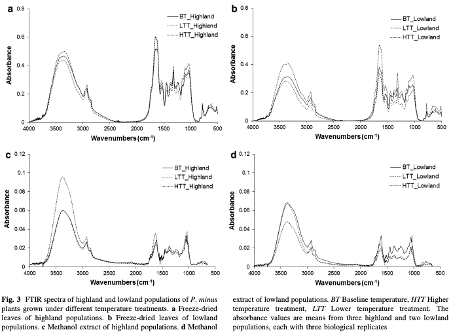
Fourier transform infrared spectroscopy was used to characterise highland and lowland populations of Polygonum minus Huds. grown in different controlled environments. A thermal perturbation technique of two-dimensional correlation infrared spectroscopy (2D-IR) correlation spectra was applied to establish differences between the populations.
Fourier transform infrared spectroscopy was used to characterise highland and lowland populations of Polygonum minus Huds. grown in different controlled environments. A thermal perturbation technique of two-dimensional correlation infrared spectroscopy (2D-IR) correlation spectra was applied to establish differences between the populations.
|
The absorption peaks at 3,480 cm−1 (hydroxyl group), 2,927 cm−1 (methyl group), 1,623 cm−1 (carbonyl group), and 1,068 cm−1 (C–O group) were particularly powerful in separating the populations. |
These peaks, which indicate the presence of carbohydrate, terpenes, amide and flavonoids were more intense for the highland populations than lowland populations, and increased in environments with a higher temperature. Wavenumbers (1,634, 669 cm−1) and (1,634, 1,555 cm−1) in the 2D-IR correlation spectra provided fingerprint signals to differentiate plants grown at different temperatures.
|
Significance
FTIR is a rapid screening method that can be applied to identify the chemical compositional difference between plant samples. P. minus plants from different populations which were subjected to different temperature treatments can be discriminated by IR spectra according to the position and intensity of peaks, based on the tri-step identification+ IR, secondary derivative-IR and 2D-IR correlation spectra. In the temperature treatment experiments, the higher temperature treatment resulted in signs of increasing carbohydrate, terpenes, amide and flavonoids, which were apparent in highland populations of both freeze-dried and flavonoid samples. Hence, the production of desired compounds could potentially be boosted by controlling the temperature conditions of plant cultivation. Terpenes and flavonoids are the major compounds in P. minus, as it could be assigned as a marker for the temperature changes especially in higher temperature. Furthermore, we can conclude that highland P. minus plants generally produced certain secondary metabolites in greater abundance than that of lowland plants.
From exploratory systems biology to translational synthetic biology
|
Functional characterisation of new sesquiterpene synthase from the Malaysian herbal plant, Polygonum minus
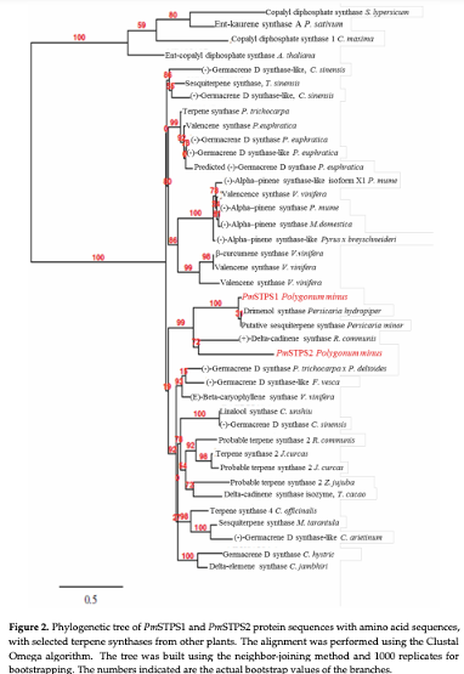
This study describes the cloning and functional characterisation of PmSTPS1 and PmSTPS2, two sesquiterpene synthase genes that were identified from P. minus transcriptome data mining. The full-length sequences of the PmSTPS1 and PmSTPS2 genes were expressed in the E. coli pQE-2 expression vector. The sizes of PmSTPS1 and PmSTPS2 were 1,098 bp and 1,967 bp, respectively, with open reading frames (ORF) of 1,047 and 1,695 bp and encoding polypeptides of 348 and 564 amino acids, respectively. The proteins consist of three conserved motifs, namely, Asp-rich substrate binding (DDxxD), metal binding residues (NSE/DTE), and cytoplasmic ER retention (RxR), as well as the terpene synthase family N-terminal domain and C-terminal metal-binding domain. From the in vitro enzyme assays, using the farnesyl pyrophosphate (FPP) substrate, the PmSTPS1 enzyme produced multiple acyclic sesquiterpenes of β-farnesene, α-farnesene, and farnesol, while the PmSTPS2 enzyme produced an additional nerolidol as a final product. The results confirmed the roles of PmSTPS1 and PmSTPS2 in the biosynthesis pathway of P. minus, to produce aromatic sesquiterpenes. |
Significance
Two new sesquiterpene synthases, PmSTPS1 and PmSTPS2, which were identified from P. minus leaf transcriptomics analysis, were cloned and characterised. Both of the enzymes produced industrially important acyclic sesquiterpenes, β-farnesene, α-farnesene, and farnesol. PmSTPS2 also produced nerolidol as the major product from FPP conversion. This study demonstrated the production of P. minus characteristic fragrance-related sesquiterpenes, by both PmSTPS1 and PmSTP, as well as the potential of further metabolic engineering in E. coli, using PmSTPS2 for the microbial production of nerolidol.
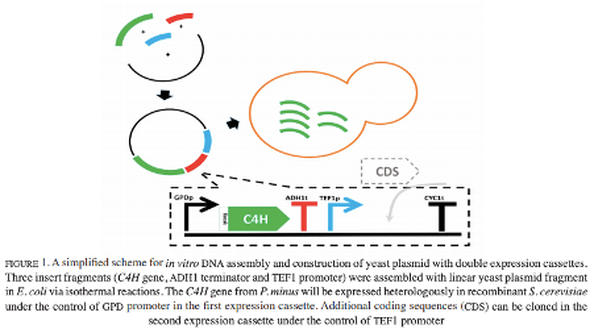
Rapid assembly of yeast expression cassettes for phenylpropanoid biosynthesis in Saccharomyces cerevisiae
Microbial production of natural products using metabolic engineering and synthetic biology approaches often involves the assembly of multiple gene fragments including regulatory elements, especially when using eukaryotes as hosts. Traditional cloning strategy using restriction enzyme digestion and ligation are laborious and inflexible owing to the high number of sequential cloning steps, limited cutting sites and generation of undesired ‘scar’ sequences.
In this study, a homology-based isothermal DNA assembly method was carried out for one-step simultaneous assembly of multiple DNA fragments to engineer plant phenylpropanoid biosynthesis in Saccharomyces cerevisiae. Rapid construction of yeast plasmid harboring dual gene expression cassettes was achieved via isothermal assembly of four DNA fragments designed with 20 bp overlapping sequences. The rate-limiting enzyme of phenylpropanoid pathway, cinnamate 4-hydroxylase encoded by C4H gene from Polygonum minus was cloned in tandem with yeast promoter and terminator elements of S. cerevisiae for efficient construction of phenylpropanoid biosynthetic pathway in recombinant yeast. The assembled pAGCAT (C4H-ADH1t-TEF1p) shuttle plasmid and transformation of S. cerevisiae with the plant C4H gene were confirmed via PCR analysis. Based on these findings, the yeast shuttle plasmid harboring P. minus phenylpropanoid biosynthesis gene was efficiently constructed to be the starting platform for the production of plant natural products in genetically engineered S. cerevisiae.
Microbial production of natural products using metabolic engineering and synthetic biology approaches often involves the assembly of multiple gene fragments including regulatory elements, especially when using eukaryotes as hosts. Traditional cloning strategy using restriction enzyme digestion and ligation are laborious and inflexible owing to the high number of sequential cloning steps, limited cutting sites and generation of undesired ‘scar’ sequences.
In this study, a homology-based isothermal DNA assembly method was carried out for one-step simultaneous assembly of multiple DNA fragments to engineer plant phenylpropanoid biosynthesis in Saccharomyces cerevisiae. Rapid construction of yeast plasmid harboring dual gene expression cassettes was achieved via isothermal assembly of four DNA fragments designed with 20 bp overlapping sequences. The rate-limiting enzyme of phenylpropanoid pathway, cinnamate 4-hydroxylase encoded by C4H gene from Polygonum minus was cloned in tandem with yeast promoter and terminator elements of S. cerevisiae for efficient construction of phenylpropanoid biosynthetic pathway in recombinant yeast. The assembled pAGCAT (C4H-ADH1t-TEF1p) shuttle plasmid and transformation of S. cerevisiae with the plant C4H gene were confirmed via PCR analysis. Based on these findings, the yeast shuttle plasmid harboring P. minus phenylpropanoid biosynthesis gene was efficiently constructed to be the starting platform for the production of plant natural products in genetically engineered S. cerevisiae.
|
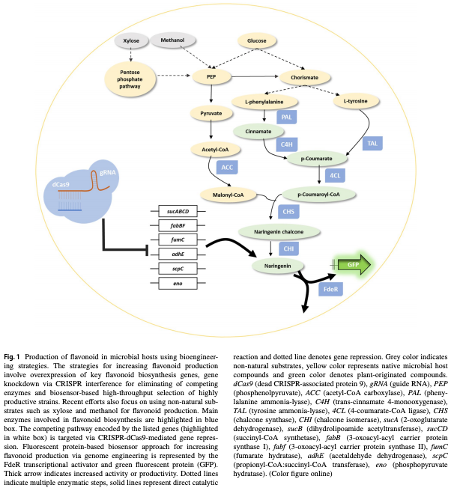
Recent advancement of engineering microbial hosts for the biotechnological production of flavonoids
Flavonoids are polyphenols that are important organic chemicals in plants. The health benefits of flavonoids that result in high commercial values make them attractive targets for large-scale production through bioengineering. Strategies such as engineering a flavonoid biosynthetic pathway in microbial hosts provide an alternative way to produce these beneficial compounds. Escherichia coli, Saccharomyces cerevisiae, and Streptomyces sp. are among the expression systems used to produce recombinant products, as well as for the production of flavonoid compounds through various bioengineering approaches including clustered regularly interspaced short palindromic repeats (CRISPR)-based genome engineering and genetically encoded biosensors to detect flavonoid biosynthesis. This article reviews the recent advances in engineering model microbial hosts as being the factory to produce targeted flavonoid compounds. |
References
Book
Grants
- Rahnamaie-Tajadod R, Goh H-H* & Normah MN (2019) Methyl jasmonate-induced compositional changes of volatile organic compounds in Polygonum minus Leaves. Journal of Plant Physiology 240, 152994.
- Rusdi NA, Goh H-H, Sabri S, Ramzi AB, Noor NM & Baharum SN (2018) Functional characterisation of new sesquiterpene synthase from the Malaysian herbal plant, Polygonum minus. Molecules 23(6):1370.
- Rahnamaie-Tajadod, R., Loke, K. K., Goh, H.H. & Noor, N. M. (2017) Differential Gene Expression Analysis in Polygonum minus Leaf Upon 24h of Methyl Jasmonate Elicitation. Frontiers in Plant Science 8:109.
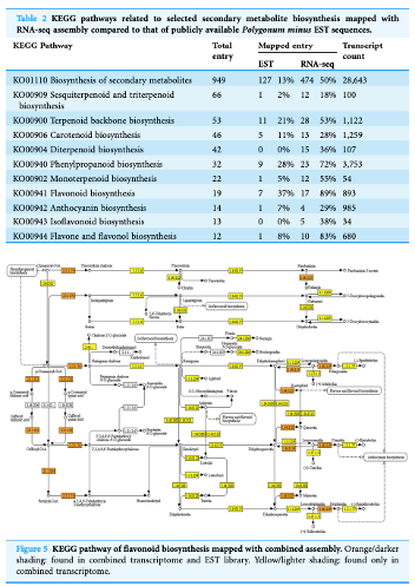
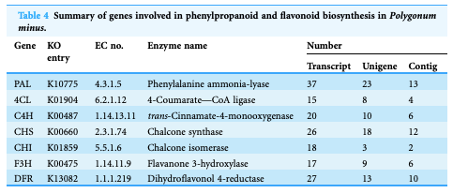
- Loke K-K, Rahnamaie-Tajadod R, Yeoh C-C, Goh H-H*, Mohamed-Hussein Z-A, Zainal Z, Ismail I. & Normah MN (2017) Transcriptome analysis of Polygonum minus reveals candidate genes involved in important secondary metabolic pathways of phenylpropanoids and flavonoids. PeerJ, 5, e2938; doi:10.7717/peerj.2938
- Aizat WM, Ibrahim S, Rahnamaie-Tajadod R, Loke K-K, Goh H-H & Noor NM (2018) Proteomics (SWATH-MS) informed by transcriptomics approach of tropical herb Persicaria minor leaves upon methyl jasmonate elicitation. PeerJ, 6:e5525.
- Aizat WM, Ibrahim S, Rahnamaie-Tajadod R, Loke KK, Goh H-H & Noor NM (2018) Extensive mass spectrometry proteomics data of Persicaria minor herb upon methyl jasmonate treatment. Data in Brief 16:1091-1094.
- Rusdi NA, Goh H-H & Baharum SN (2016) GC-MS/Olfactometric characterisation and aroma extraction dilution analysis of aroma active compounds in Polygonum minus essential oil. Plant Omics Journal, 9(4), 289-294.
- Loke K-K, Rahnamaie-Tajadod R, Yeoh C-C, Goh H-H*, Mohamed-Hussein Z-A, Normah M.N., Zainal Z & Ismail I. (2016) RNA-seq analysis for secondary metabolite pathway gene discovery in Polygonum minus. Genomics Data, 7, 12-13.
- Goh H-H, Khairudin K, Sukiran N, Baharum S & Normah M.N. (2016) Metabolite profiling reveals temperature effects on the VOCs and flavonoids of different plant populations. Plant Biology, 18(1), 130-139. PDF
- Khairudin K, Sukiran N, Goh H-H, Baharum S. & Normah M.N. (2014). Direct discrimination of different plant populations and study on temperature effects by Fourier transform infrared spectroscopy. Metabolomics, 10, 203–211, doi:10.1007/s11306-013-0570-5 PDF
- Ramzi AB, Ku Bahaudin KNA, Baharum SN, Che Me ML, Goh H-H, Hassan M & Noor NM (2018) Rapid assembly of yeast expression cassettes for phenylpropanoid biosynthesis in Saccharomyces cerevisiae. Sains Malaysiana 47(12):2969-2974.
- Shah FLA, Ramzi AB, Baharum SN, Noor NM, Goh H-H, Leow TC, Oslan SN & Sabri S (2019) Recent advancement of engineering microbial hosts for the biotechnological production of flavonoids. Molecular Biology Reports 46:6647–6659.
Book
- Sukiran N.A., Goh H-H. & Baharum S. N. (2018) Kesan altitud dan suhu terhadap penghasilan sebatian kimia dalam Polygonum minus. UKM Press ISBN 9789674127343
Grants
- Establishing the involvement of a kelch repeat/F-Box protein (KFB) in oxylipin pathway for the production of green leaf volatiles - FRGS/1/2015/SG03/UKM/01/1 [2 Nov 2015 - 1 Nov 2018] – Co-researcher
- EXPLORATION OF POLYGONUM MINUS FRAGRANCE USING HYPHENATED GC-MS-ELECTRONIC-NOSE (E-NOSE) TECHNOLOGY FOR NOVEL SCENT GENE(S) Research University Grant: GUP-2013-028 [1 Oct 2013 – 30 Sep 2015] – Co-researcher
- APPLICATION OF STATE-OF-ART DEGRADOME TECHNOLOGY TO DETERMINE AND ANALYSE THE REGULATORY MICRO RNAS (MIRNAS) OF FLAVONOID BIOSYNTHESIS PATHWAY IN POLYGONUM MINUS - FRGS/1/2013/SG05/UKM/01/2 [1 Apr 2013 – 31 Mar 2015] – Co-researcher
- METABOLIC ENGINEERING FLAVONOID PRODUCTION IN YEAST - MOSTI-ASM: PKA0514F004 [1 Sep 2015 – 28 Feb 2018] – Co-researcher
- INCREASING FLAVONOID PRODUCTION IN LACTOCOCCUS LACTIS THROUGH METABOLIC FLUX ANALYSIS - SCIENCEFUND MOSTI: 02-01-02-SF0987 [1 Jun 2013 – 31 Mar 2016] – Co-researcher
- SYSTEMIC CHARACTERISATION AND MANIPULATION OF PHENYLPROPANOID PATHWAY USING MICROBIAL SYSTEMS FOR THE PRODUCTION OF FINE CHEMICALS FROM LIGNIN - Research University Grant: AP-2013-013 [1 Sep 2013 – 29 Feb 2016] – Co-researcher
Copyright © 2022 UKM